Carbohydrates are good source of energy and a long chain of sugar. It consist of three classes that is monosaccharides, disaccharides and polysaccharides. Carbohydrates are found in a wide array of both healthy and unhealthy foods—bread, beans, milk, popcorn, potatoes, cookies, spaghetti, soft drinks, corn, and cherry pie. They also come in a variety of forms. The most common and abundant forms are sugars, fibers, and starches.Foods high in carbohydrates are an important part of a healthy diet. Carbohydrates provide the body with glucose, which is converted to energy used to support bodily functions and physical activity. But carbohydrate quality is important; some types of carbohydrate-rich foods are better than others:
MONOSACCHARIDES
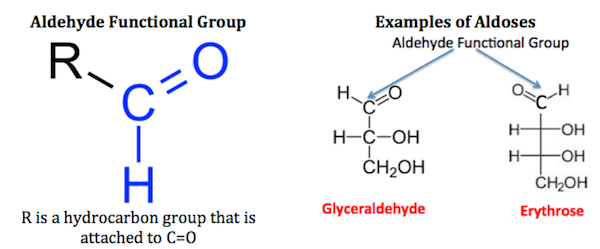
Monosaccharides are classified as well based on their functional group. A functional group are categorized by atoms or bonds that are responsible for the chemical activity within the molecule. The functional group include: Ketose and aldose.
Ketose Group:
- The healthiest sources of carbohydrates—unprocessed or minimally processed whole grains, vegetables, fruits and beans—promote good health by delivering vitamins, minerals, fiber, and a host of important phytonutrients.
- Unhealthier sources of carbohydrates include white bread, pastries, sodas, and other highly processed or refined foods. These items contain easily digested carbohydrates that may contribute to weight gain, interfere with weight loss, and promote diabetes and heart disease.
MONOSACCHARIDES
- The simplest unit of carbohydrates and the simplest form of sugar.
- The building blocks of more complex carbohydrates that is disaccharides and polysaccharides.
- Usually colourless, can dissolve in water, and have the appearance of crystal-like substance.
- Example: Glucose, Fructose.
- Formula: (CH20)n , n ≥ 3
Monosaccharides are classified as well based on their functional group. A functional group are categorized by atoms or bonds that are responsible for the chemical activity within the molecule. The functional group include: Ketose and aldose.
Ketose Group:
- If a monosaccharides contain a ketone group in the inner atom, then the monosaccharides are classified as ketone group.
- Ketone group is a carbon atom forming double bond with the oxygen atom and single bond with the hydrocarbon groups ( group that contain carbon bonded with hydrogen).
- Below is the general appearance of ketose group:
Aldose Group:
- Monosaccharides that contain aldehyde group at an end carbon.
- Aldehyde group is the carbon atom forming a double bond with the oxygen atom and single bond with hydrogen.
- Below is the general appearance of aldose group.
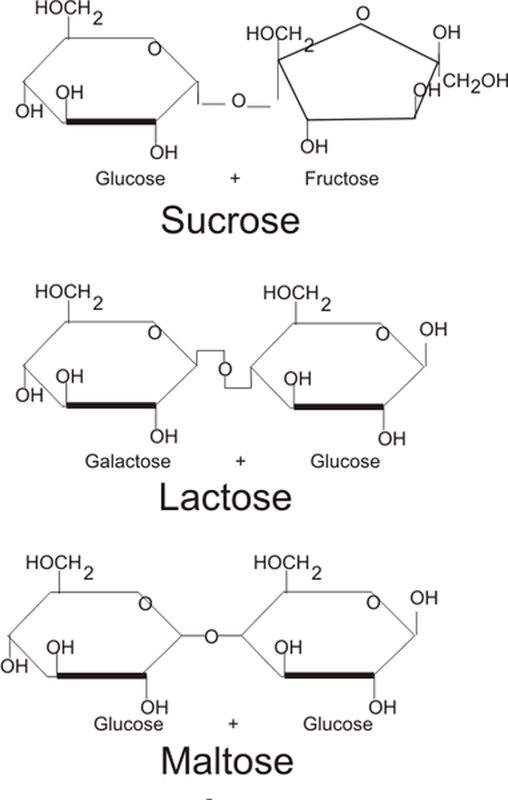
DISACCHARIDES
Formation of disaccharide sucrose from monosaccharides fructose and glucose.
- Also called double sugar.
- Composed of two monosaccharides (simple sugar) linked to each other.
- Crystalline water-soluble compound.
- The monosaccharides linked within them by gycosidic bond.
- The monosaccharides joined by the process of dehydration synthesis as shown below:
Formation of disaccharide sucrose from monosaccharides fructose and glucose.
- Disaccharides also know as complex sugar.
- The sugar are created by through dehydration synthesis and broken down by hydrolysis.
- The common disaccharides are sucrose, lactose and maltose.
POLYSACCHARIDES
- A long chain of monosaccharides that linked by glycosidic bond.
- Typical polysaccharides is between 200 and 2500 monosaccharides long.
- Can be either linear or branch carbon chain.
- Usual structure of polysaccharides consist of 6 carbon monosaccharides linked by oxygen.
- Formula: ( C6H10O5)n, n > 40.
- Example of polysaccharides: Amylose, Glycogen.
- Can be used as energy storage.
- Insoluble in water and not in crystalline form.
- Structure of polysaccharide shown as below:
Typical structure of polysaccharides
properties of carbohydratesGeneral properties of carbohydrates:
Physical properties of carbohydrates:
Chemical properties of carbohydrates:
properties of carbohydratesGeneral properties of carbohydrates:
- Carbohydrates act as energy reserves, also stores fuels, and metabolite intermediate.
- Ribose and deoxyribose form the structural frame of genetic material, RNA and DNA.
- Structural element in the cell wall of bacteria and plant.
Physical properties of carbohydrates:
- Steroisomerism - Compound shaving same structural formula but they differ in spatial configuration. Example: Glucose has two isomers with respect to penultimate carbon atom. They are D-glucose and L-glucose.
- Optical Activity - It is the rotation of plane polarized light forming (+) glucose and (-) glucose.
- Diastereo isomeers - It the configurational changes with regard to C2, C3, or C4 in glucose. Example: Mannose, galactose.
- Annomerism - It is the spatial configuration with respect to the first carbon atom in aldoses and second carbon atom in ketoses.
Chemical properties of carbohydrates:
- Ozazone formation with phenylhydrazine.
- Benedicts test.
- Oxidation
- Reduction to alcohols
Protein structure is organized into four different levels. Another way to say this, is that proteins have four levels of structure. Let’s start out with the most basic level: primary structure.
Primary Structure of Proteins
The primary structure of a protein is simply its amino acid sequence. If you recall, amino acids are the subunits that make up proteins. This level of structure is similar to letters that are strung together to create a word or beads on a string
Primary Structure of Proteins
The primary structure of a protein is simply its amino acid sequence. If you recall, amino acids are the subunits that make up proteins. This level of structure is similar to letters that are strung together to create a word or beads on a string
Secondary Structure of Proteins
Secondary structure is when intermolecular forces, specifically hydrogen bonds between the amine and carboxyl groups of amino acids, cause the primary sequence to bend or twist. A bend is called a β-pleated sheet and looks similar to apaper fan. Twists are called alpha helices and look like an old telephone cord, like the one your mom might have had in the kitchen.
Secondary structure is when intermolecular forces, specifically hydrogen bonds between the amine and carboxyl groups of amino acids, cause the primary sequence to bend or twist. A bend is called a β-pleated sheet and looks similar to apaper fan. Twists are called alpha helices and look like an old telephone cord, like the one your mom might have had in the kitchen.
Tertiary Structure of Proteins
When the primary sequence and secondary sequences bend and fold upon themselves, we call this the tertiary structure. This third level of structure is caused by intermolecular forces (such as hydrophobic interactions or hydrogen bonding), ionic bonding and covalent disulfide bridges. Unlike in the secondary structure where the structure is formed from interactions between the polypeptide backbone (the amine – carboxyl groups), these interactions in the tertiarystructure are driven by the characteristics of the R-groups. Tertiary structure can be visualized again by your kitchen telephone cord. Imagine the alpha helix of the cord twisted, so that now we have a twisted alpha helix. This is the tertiary structure of a polypeptide.
When the primary sequence and secondary sequences bend and fold upon themselves, we call this the tertiary structure. This third level of structure is caused by intermolecular forces (such as hydrophobic interactions or hydrogen bonding), ionic bonding and covalent disulfide bridges. Unlike in the secondary structure where the structure is formed from interactions between the polypeptide backbone (the amine – carboxyl groups), these interactions in the tertiarystructure are driven by the characteristics of the R-groups. Tertiary structure can be visualized again by your kitchen telephone cord. Imagine the alpha helix of the cord twisted, so that now we have a twisted alpha helix. This is the tertiary structure of a polypeptide.
Quaternary Structure of Proteins
The final level of protein structure is quaternary structure. Quaternary structure when multiple polypeptides interact with each other to fold. This is similar to maybe a box of computer cables tangled in a box in your closet or multiple telephone cords, each twisted upon the alpha helices and then all tangled together. The types of forces that make up quaternary structure are the same that are found at the tertiary level: intermolecular forces such as hydrophobic interactions or hydrogen bonding, ionic bonding and covalent disulfide bridges. In the drawing below, each color represents individual polypeptide chains. This protein is made up of 4 different polypeptide chains, red, green, black and blue.
The final level of protein structure is quaternary structure. Quaternary structure when multiple polypeptides interact with each other to fold. This is similar to maybe a box of computer cables tangled in a box in your closet or multiple telephone cords, each twisted upon the alpha helices and then all tangled together. The types of forces that make up quaternary structure are the same that are found at the tertiary level: intermolecular forces such as hydrophobic interactions or hydrogen bonding, ionic bonding and covalent disulfide bridges. In the drawing below, each color represents individual polypeptide chains. This protein is made up of 4 different polypeptide chains, red, green, black and blue.
Lipids
Lipids are important constituent of of the diet because they are a source of high energy value. Lipids are also important because of the fat-soluble vitamins, and essential fatty acids found in the fat of the natural food stuffs. Body fat serves as a very good source of energy, it is stored in adipose tissues. They also act as insulating material in the subcutaneous tissues and are also seen around certain organs. Lipids combined with proteins are important constituents of the cell membranes and mitochondria of the cell. Lipids are not generally macromolecules.
Lipids are naturally occurring organic compounds, commonly known as oils and fats. Lipids occur through out the living world in microorganisms, higher plants and animals and also in all cell types. Lipids contribute to cell structure, provide stored fuel and also take part in many biological processes.
Lipids are important constituent of of the diet because they are a source of high energy value. Lipids are also important because of the fat-soluble vitamins, and essential fatty acids found in the fat of the natural food stuffs. Body fat serves as a very good source of energy, it is stored in adipose tissues. They also act as insulating material in the subcutaneous tissues and are also seen around certain organs. Lipids combined with proteins are important constituents of the cell membranes and mitochondria of the cell. Lipids are not generally macromolecules.
Lipids are naturally occurring organic compounds, commonly known as oils and fats. Lipids occur through out the living world in microorganisms, higher plants and animals and also in all cell types. Lipids contribute to cell structure, provide stored fuel and also take part in many biological processes.
General characters of lipids are
- Lipids are relatively insoluble in water.
- They are soluble in non-polar solvents, like ether, chloroform, methanol.
- Lipids have high energy content and are metabolized to release calories.
- Lipids also act as electrical insulators, they insulate nerve axons.
- Fats contain saturated fatty acids, they are solid at room temperatures. Example, animal fats.
- Plant fats are unsaturated and are liquid at room temperatures.
- Pure fats are colorless, they have extremely bland taste.
- The fats are sparingly soluble in water and hence are described are hydrophobic substances.
- They are freely soluble in organic solvents like ether, acetone and benzene.
- The melting point of fats depends on the length of the chain of the constituent fatty acid and the degree of unsaturation.
- Geometric isomerism, the presence of double bond in the unsaturated fatty acid of the lipid molecule produces geometric or cis-trans isomerism.
- Fats have insulating capacity, they are bad conductors of heat.
- Emulsification is the process by which a lipid mass is converted to a number of small lipid droplets. The process of emulsification happens before the fats can be absorbed by the intestinal walls.
- The fats are hydrolyzed by the enzyme lipases to yield fatty acids and glycerol.
- The hydrolysis of fats by alkali is called saponification. This reaction results in the formation of glycerol and salts of fatty acids called soaps.
- Hydrolytic rancidity is caused by the growth of microorganisms which secrete enzymes like lipases. These split fats into glycerol and free fatty acids.
imple Lipids or Homolipids
Simple lipids are the esters of fatty acids with various alcohols.
Fats and Oils (triglycerides and triacylglycerols) - These are esters of fatty acids with a trihydroxy alcohol, glycerol. A fat is solid at ordinary room temperature, an oil is liquid.
Simple Triglycerides - Simple triglycerides are one in which three fatty acids radicles are similar or are of the same type. Example: Tristearin, Triolein.
Mixed Triglycerides are one in which the three fatty acids radicles are different from each other. Example: distearo-olein, dioleo-palmitin.
Simple lipids are the esters of fatty acids with various alcohols.
Fats and Oils (triglycerides and triacylglycerols) - These are esters of fatty acids with a trihydroxy alcohol, glycerol. A fat is solid at ordinary room temperature, an oil is liquid.
Simple Triglycerides - Simple triglycerides are one in which three fatty acids radicles are similar or are of the same type. Example: Tristearin, Triolein.
Mixed Triglycerides are one in which the three fatty acids radicles are different from each other. Example: distearo-olein, dioleo-palmitin.
Waxes are the esters of fatty acids with high molecular weight monohydroxy alcohols. Example: Beeswax, Carnauba wax.
Compound Lipids or Heterolipids
Heterolipids are esters of fatty acids with alcohol and possess additional groups also.
Phospholipids or Phosphatids are compound containing fatty acids and glycerol in addition to a phosphoric acid, nitrogen bases and other substituents. They usually possess one hydrophilic head and tow non-polar tails. They are called polar lipids and are amphipathic in nautre.
Phospholipids can be phosphoglycerides, phosphoinositides and phosphosphingosides.
Phosphoglycerides are major phospholipids, they are found in membranes. It contains fatty acid molecules which are esterified to hydroxyl groups of glycerol. The glycerol group also forms an ester linkage with phosphoric acid. Example: Lecithin, Cephalins.
Phosphoinositides are said to occur in phospholipids of brain tissue and soybeans. The ply important role in transport processes in cells.
Compound Lipids or Heterolipids
Heterolipids are esters of fatty acids with alcohol and possess additional groups also.
Phospholipids or Phosphatids are compound containing fatty acids and glycerol in addition to a phosphoric acid, nitrogen bases and other substituents. They usually possess one hydrophilic head and tow non-polar tails. They are called polar lipids and are amphipathic in nautre.
Phospholipids can be phosphoglycerides, phosphoinositides and phosphosphingosides.
Phosphoglycerides are major phospholipids, they are found in membranes. It contains fatty acid molecules which are esterified to hydroxyl groups of glycerol. The glycerol group also forms an ester linkage with phosphoric acid. Example: Lecithin, Cephalins.
Phosphoinositides are said to occur in phospholipids of brain tissue and soybeans. The ply important role in transport processes in cells.
Phosphosphingosides are commonly found in nerve tissue. Example: sphingomyelins.
Glycolipids are the compounds of fatty acids with carbohydrates and contain nitrogen but no phosphoric acid. The glycolipids also include certain structurally related compounds comprising the groups gangliosides, sulpholipids and sulfatids.
Derived Lipids
Derived lipids are the substances derived from simple and compound lipids by hydrolysis. These includes fatty acids, alcohols, monoglycerides and diglycerides, steroids, terpenes, carotenoids.
The most common derived lipids are steroids, terpenes and carotenoids.
Steroids do not contain fatty acids, they are nonsaponifiable, and are not hydrolyzed on heating. They are widely distributed in animals, where they are associated with physiological processes. Example: Estranes, androstranes, etc.
Glycolipids are the compounds of fatty acids with carbohydrates and contain nitrogen but no phosphoric acid. The glycolipids also include certain structurally related compounds comprising the groups gangliosides, sulpholipids and sulfatids.
Derived Lipids
Derived lipids are the substances derived from simple and compound lipids by hydrolysis. These includes fatty acids, alcohols, monoglycerides and diglycerides, steroids, terpenes, carotenoids.
The most common derived lipids are steroids, terpenes and carotenoids.
Steroids do not contain fatty acids, they are nonsaponifiable, and are not hydrolyzed on heating. They are widely distributed in animals, where they are associated with physiological processes. Example: Estranes, androstranes, etc.
Terpenes in majority are found in plants. Example: Natural rubber. gernoil, etc.
Carotenoids are tetraterpenes. They are widely distributed in both plants and animals. They are exclusively of plant origin. Due to the presence of many conjugated double bonds, they are colored red or yellow. Example: Lycopreene, carotenes, Xanthophylls.
Essential fatty acids are those that cannot be constructed through any chemical pathways, known to happen in humans. They must be obtained from the diet. Linoleic acid and linolenic acid are the essential fatty acids.
Non-essential fatty acids are those which are not necessary to be taken through diet, they are synthesized through chemical pathways.
Unsaturated fatty acids have one or more double bonds between carbon atoms. The tow carbon atoms are bound to each other through double bonds and can occur in cis or trans configuration.
Saturated fatty acids are long chain carboxylic acids and do not have double bonds. Example: Arachidic acid, Palmitic acid, etc.
Structure of Lipids
Lipids has no single common structure. The most commonly occurring lipids are triglycerides and phospholipids.
Triglycerides are fats and oils. Triglycerides have a glycerol backbone bonded to three fatty acids. If the three fatty are similar then the triglyceride is known as simple triglyceride. If the fatty acids are not similar then the fatty acids are known as mixed triglyceride.
The second most common class of lipids are phospholipids. They are found in membranes of animal and plants. Phospholipids contains glycerol and fatty acids, they also contain phosphoric acids and a low-molecular weight alcohol. Common phospholipids are lecithins and cephalins.
Function of Lipids
Lipids perform several biological functions:
Carotenoids are tetraterpenes. They are widely distributed in both plants and animals. They are exclusively of plant origin. Due to the presence of many conjugated double bonds, they are colored red or yellow. Example: Lycopreene, carotenes, Xanthophylls.
Essential fatty acids are those that cannot be constructed through any chemical pathways, known to happen in humans. They must be obtained from the diet. Linoleic acid and linolenic acid are the essential fatty acids.
Non-essential fatty acids are those which are not necessary to be taken through diet, they are synthesized through chemical pathways.
Unsaturated fatty acids have one or more double bonds between carbon atoms. The tow carbon atoms are bound to each other through double bonds and can occur in cis or trans configuration.
Saturated fatty acids are long chain carboxylic acids and do not have double bonds. Example: Arachidic acid, Palmitic acid, etc.
Structure of Lipids
Lipids has no single common structure. The most commonly occurring lipids are triglycerides and phospholipids.
Triglycerides are fats and oils. Triglycerides have a glycerol backbone bonded to three fatty acids. If the three fatty are similar then the triglyceride is known as simple triglyceride. If the fatty acids are not similar then the fatty acids are known as mixed triglyceride.
The second most common class of lipids are phospholipids. They are found in membranes of animal and plants. Phospholipids contains glycerol and fatty acids, they also contain phosphoric acids and a low-molecular weight alcohol. Common phospholipids are lecithins and cephalins.
Function of Lipids
Lipids perform several biological functions:
- Lipids are storage compounds, triglycerides serve as reserve energy of the body.
- Lipids are important component of cell membranes structure in eukaryotic cells.
- Lipids regulate membrane permeability.
- They serve as source for fat soluble vitamins like A, D, E, K.
- They act electrical insulators to the nerve fibres, where the myelin sheath contains lipids.
- Lipids are components of some enzyme systems.
- Some lipids like prostaglandins and steroid hormones act as cellular metabolic regulators.
- Cholesterol is found in cell membranes, blood, and bile of many organisms.
- As lipids are small molecules and are insoluble in water, they act as signalling molecules.
- Layers of fat in the subcutaneous layer, provides insulation and protection from cold. Body temperature maintenance is done by brown fat.
- Polyunsaturated phospholipids are important constituents of phospholipids, they provide fluidity and flexibility to the cell membranes.
- Lipoproteins that are complexes of lipids and proteins, occur in blood as plasma lipoprotein, they enable transport of lipids in aqueous environment, and their transport throughout the body.
- Cholesterol maintains fluidity of membranes by interacting with lipid complexes.
- Cholesterol is the precursor of bile acids, Vitamin D and steroids.
- Essential fatty acids like linoleic and linolenic acids are precursors of many different types of ecosanoids including prostaglandins, thromboxanes. These play a important role in pain, fever, inflammation and blood clotting.
Nucleic Acids
Nucleic acids are large organic compounds found in the chromosomes of living cells and viruses. They are strong acids found in the nucleus of the cells. The nucleic acid polymers are with high molecular weights as high as 100,000,000 grams per mole. With proteins, nucleic acids are most important biological macromolecules. They are found in abundance in all living cells.
DefinitionNucleic acids are essential large biological molecules for all forms of life. The nucleic acids include the DNA and the RNA. They are the hereditary determinants of living organisms. They are present in most living cells either in free state or bound to proteins as nucleoproteins. The nucleic acids are biopolymers with mononucleotides ad their repeating units. The monomers are known as nucleotides, they are made up of three units: a sugar, an amine and a phosphate group.
Nucleic acids are large organic compounds found in the chromosomes of living cells and viruses. They are strong acids found in the nucleus of the cells. The nucleic acid polymers are with high molecular weights as high as 100,000,000 grams per mole. With proteins, nucleic acids are most important biological macromolecules. They are found in abundance in all living cells.
DefinitionNucleic acids are essential large biological molecules for all forms of life. The nucleic acids include the DNA and the RNA. They are the hereditary determinants of living organisms. They are present in most living cells either in free state or bound to proteins as nucleoproteins. The nucleic acids are biopolymers with mononucleotides ad their repeating units. The monomers are known as nucleotides, they are made up of three units: a sugar, an amine and a phosphate group.
Properties of nucleic acids
Types of Nucleic AcidsNucleic acids are of two types DNA and RNA
- Nucleic acids (DNA and RNA) are long polymers made of repeating units of nucleotides.
- Nucleotide units are made of phosphate-sugar-nitrogenous base units.
- The nitrogenous bases found in DNA are adenine, guanine, cytosine and thymine.
- Adenine and guanine are purine bases while cytosine and thymine are pyrimidnes.
- In RNA, the thymine bases id replaced by uracil which is also a pyrimidine.
- The nucleotides are linked with phosphodiester bonds.
- They are linked by a phosphate group on the 5th position of sugar residue becomes linked to 3' hydroxyl group of the preceeding sugar molecule.
- The double stranded model of the DNA was worked out by Watson and Crick in 1953.
- The double helix model consists of two strands wound around a central axis with the bases stacked inside.
- The order of the strand is in opposite directions, i.e., from 5' to 3' direction in one and 3'to 5' direction in the other.
- The bases stacked in the center of the helix as they interact with each other through weak hydrogen bonds.
- Hydrogen bonds are weaker than covalent bonds, they are continually forming and disassociating.
- In the double stranded nucleic acids, the adenine form hydrogen bonds only with thymine (or uracil) molecule. While cytidine will only form hydrogen bonds with guanine.
- Hence, in a a given strand of DNA, the amount of adenine is always equal to the amount of thymine, and the amount of cytidine always equals the amount of guanine, in a given species.
- The per cent of the G-T and the A-T is variable from species to species.
- The base pairs form a flat plain in the helix, the adenine forming two hydrogen bonds with thymine, and the cytidine forming three bonds with guanine.
- Using the concept of base pairing, all the enzymes and substrates necessary, the two DNA strands when copied separately, wherever there is adenine in the original strand, the duplicated strand will have thymidine, and guanine would be matched to cytosine.
- After replication, each original strand (parent strand), is paired with a new (daughter) strand. This type of replication is known as semi-conservative mode of duplication.
- The double stranded DNA can be denatured by alkaline conditions or heat.
- Small stretches of polynucleotides will anneal to larger single stranded DNA molecule if the DNA sequence matches, by base pairing.
Types of Nucleic AcidsNucleic acids are of two types DNA and RNA
DNA (deoxyribonucleic acid)
- DNA is one of the macromolecules, they are essential to all living forms.
- Deoxyribonucleic acid contains the genetic information, it is used in the development and functioning of all living organisms.
- The DNA segments carry genetic information are called the genes.
- Other DNA segments have structural functions or regulate the genetic information.
- DNA are made of two chains made of polymer units of nucleotides.
- The backbones of DNA are made of sugar and phosphate groups which are joined by ester bonds.
- The two strands of DNA are anti-parallel, they run in opposite directions.
- Each sugar molecule is attached to one of the four nucleobases.
- The nucleobases encode genetic information, that is read using the genetic code.
- Inside the cell, the DNA are arranged in long structures called chromosomes.
- The chromosomes are duplicated in the process of DNA replication, during cell division.
- Each cell has its own one complete set of chromosomes.
- In eukaryotic organisms, most DNA is stored in the nucleus of the cell, and also some of it in cellular organelles like mitochondria or chloroplast.
- The prokaryotes store the DNA in the cytoplasm.
- Chromatin proteins like histones compact and organize the DNA
- The functions of ribonucleic acid is to convert genetic information from genes into amino acid sequences of protein.
- In some viruses, RNA contains the genetic information.
- RNA is of three types, they are tRNA (transfer RNA), mRNA (messenger RNA) and rRNA (ribosomal RNA).
- Messenger RNA, as the name suggests acts a messenger. It carries genetic information sequences between DNA and ribosomes, and it also directs protein synthesis.
- rRNA is a major component of the ribososmes, they catalyze the formation of peptide bond.
- The tRNA act as a carrier molecule for the amino acids that are used in protein synthesis. The tRNA are also responsible for decoding the mRNA.
Primary Structure
Primary structure of nucleic acids is a linear sequence of nucleotides, which are linked to each other by phosphodiester linkages. Nucleotides are made up of three components - Nitrogenous base, 5-carbon sugar and phosphate groups.
Nitrogenous base are purines(adenine, guanine) and pyrimidines {cytosine, thymine (present in DNA only), uracil (present in RNA only)}. The 5-carbon sugar is deoxyribose for DNA and and ribose sugar in RNA. The purine bases, form glycosidic bond between their 9' nitrogen and the 9' - OH group of the sugar molecule. The pyrimidine bases, they form glycosidic bond between 1' nitrogen and the 9' -OH of the deoxyribose. In both purine and pyrimidine bases the phosphate group forms a bond with the sugar molecule between one of its negatively charged oxygen groups and the 5' -OH of the sugar. Nucleotides forms phosphodiester linkages between the 5' and 3' carbon atoms, these form the nucleic acids. Nucleotides sequences are complementary to one another.
Example of complementary sequence AGCT is TCGA.
Primary structure of nucleic acids is a linear sequence of nucleotides, which are linked to each other by phosphodiester linkages. Nucleotides are made up of three components - Nitrogenous base, 5-carbon sugar and phosphate groups.
Nitrogenous base are purines(adenine, guanine) and pyrimidines {cytosine, thymine (present in DNA only), uracil (present in RNA only)}. The 5-carbon sugar is deoxyribose for DNA and and ribose sugar in RNA. The purine bases, form glycosidic bond between their 9' nitrogen and the 9' - OH group of the sugar molecule. The pyrimidine bases, they form glycosidic bond between 1' nitrogen and the 9' -OH of the deoxyribose. In both purine and pyrimidine bases the phosphate group forms a bond with the sugar molecule between one of its negatively charged oxygen groups and the 5' -OH of the sugar. Nucleotides forms phosphodiester linkages between the 5' and 3' carbon atoms, these form the nucleic acids. Nucleotides sequences are complementary to one another.
Example of complementary sequence AGCT is TCGA.
Secondary Structure
Secondary structure is the interaction between the bases. This structure shows parts of which strands are bound to each other. The two strands of DNA in the double helix of the DNA are bound to each other by hydrogen bounds. The nucleotides on one strand base pairs with the nucleotides of the other strand. The secondary structure of the DNA is predominantly the base pairing of the two polynucleotide strands forming a double helix.
Secondary structure is the interaction between the bases. This structure shows parts of which strands are bound to each other. The two strands of DNA in the double helix of the DNA are bound to each other by hydrogen bounds. The nucleotides on one strand base pairs with the nucleotides of the other strand. The secondary structure of the DNA is predominantly the base pairing of the two polynucleotide strands forming a double helix.
Tertiary Structure
Tertiary structure is the three dimensional shape into which the entire chain is folded. Tertiary structure arrangement differs in four structural forms:
Quaternary Structure
Quaternary structure is the higher-level of organization of the nucleic acids. This structure refers to the interactions of the nucleic acids with the other molecules. The most commonly seen organization is the form of chromatin which shows interaction with small proteins histones.
Functions of Nucleic AcidsFunctions of nucleic acids are:
Tertiary structure is the three dimensional shape into which the entire chain is folded. Tertiary structure arrangement differs in four structural forms:
- Left or right handedness.
- Length of the turn of the helix.
- Number of base pairs per turn.
- The difference in size between major and the minor groove.
Quaternary Structure
Quaternary structure is the higher-level of organization of the nucleic acids. This structure refers to the interactions of the nucleic acids with the other molecules. The most commonly seen organization is the form of chromatin which shows interaction with small proteins histones.
Functions of Nucleic AcidsFunctions of nucleic acids are:
- The main functions is store and transfer genetic information.
- To use the genetic information to direct the synthesis of new protein.
- The deoxyribonucleic acid is the storage for place for genetic information in the cell.
- DNA controls the synthesis of RNA in the cell.
- The genetic information is transmitted from DNA to the protein synthesizers in the cell.
- RNA also directs the production of new protein by transmitting genetic information to the protein building structures.
- The function of the nitrogenous base sequences in the DNA backbone determines the proteins being synthesized.
- The function of the double helix of the DNA is that no disorders occur in the genetic information if it is lost or damaged.
- RNA directs synthesis of proteins.
- m-RNA takes genetic message from RNA.
- t-RNA transfers activated amino acid, to the site of protein synthesis.
- r-RNA are mostly present in the ribosomes, and responsible for stability of m-RNA.